Saarbrücken, April 24, 2023 - On the World Day for Laboratory Animals, special attention is paid to the use of animals for research and development of products for humans. In drug research, among other areas, extensive animal testing is a firmly prescribed part of the approval process for new drugs. However, a reduction in the number of laboratory animals is increasingly in sight due to new approaches. At the Helmholtz Institute for Pharmaceutical Research Saarland (HIPS), a site of the Helmholtz Centre for Infection Research (HZI) in collaboration with Saarland University, researchers are successfully developing alternative models and have already received awards for their work.
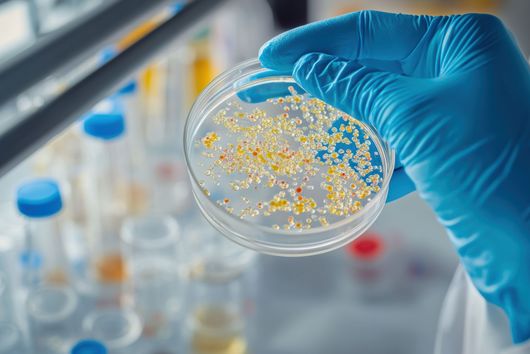
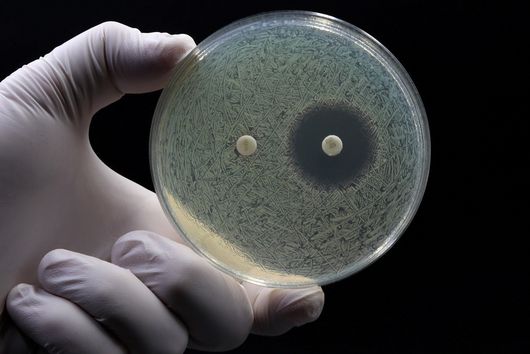
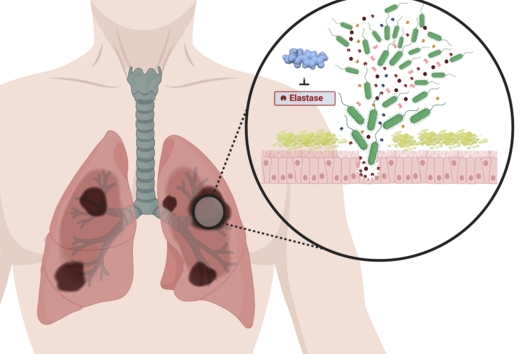
The use of animal experiments, for example on mice and other rodents, in drug research is necessary to carry out a risk assessment before a potential drug is tested on humans. This also applies, for example, to the development of new antibiotics, where it must also be ensured that the new antibiotic is safe and has no side effects. Before drugs are tested on living animals at all (in vivo studies), they have usually already undergone a large number of laboratory tests. In these so-called in vitro studies, questions are asked such as: Does the active substance interact sufficiently well with the desired target structure? Does the substance bind to other target structures that could lead to side effects? Against which pathogens does the drug candidate act? An increasing number and, above all, different in vitro experiments are available that provide a detailed picture of the mode of action of the substance. However, questions about the effect of a potential drug in humans can often only be answered in experiments on living organisms, as these involve very complex interactions and systems. These include studies that investigate the Absorption, Distribution, Metabolism, Elimination, and Toxicity of a compound. These ADME-tox studies determine exactly how a drug behaves in the organism and what the body does with the drug. Only when these aspects reveal no evidence of problems a drug candidate can be tested in clinical trials.
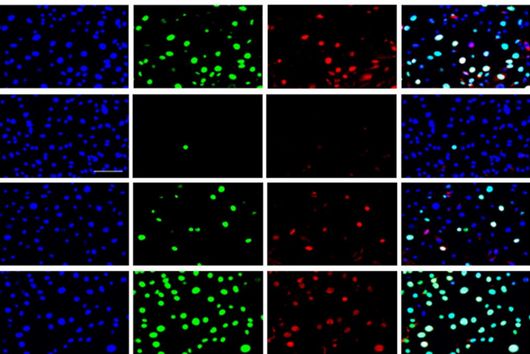
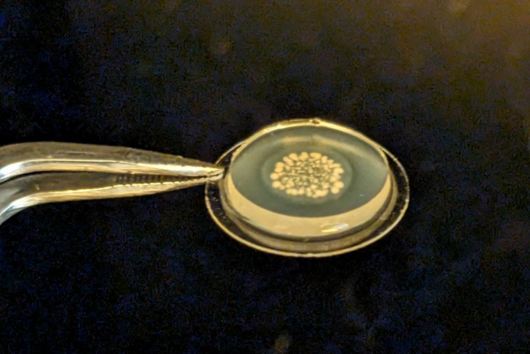
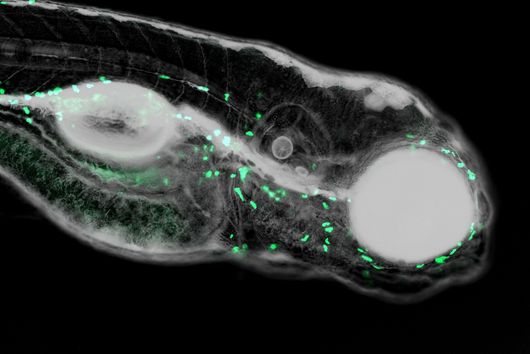
There are also new approaches for ADME tox studies that can minimize the need for animal models. One of the most promising approaches is the zebrafish larvae model (ZLM). In terms of its genetic information, zebrafish has a surprisingly high degree of similarity to humans: About 70 percent of its genes are orthologous to humans. This means that these genes are found in both organisms and have a high degree of similarity in the sequence of DNA bases. For genes with specific disease relevance, this percentage is as high as 84 percent. Dr Jonas Baumann, head of the zebrafish facility at the Helmholtz Institute for Pharmaceutical Research Saarland (HIPS), explains: "Due to the very high genetic similarity, the ZLM is ideally suited to test at a very early stage whether an active substance is degraded or shows toxic side effects." After fertilization, the larvae already form an organ system comparable to that of humans in the first 48 hours. This makes it possible to observe very precisely whether there are any abnormalities or disturbances in the development of individual organs that may be triggered by the added compound. "This allows us to test this much better than with simple cell culture models. Not only the kidneys or liver, for example, but also complex developmental abnormalities of the heart, e.g. heart beat rates and cardiac arrhythmias, and even immune responses can be detected in this way," says Baumann. According to EU guidelines, tests with fertilized zebrafish eggs are considered advanced cellular experiments up to 120 hours after fertilization of the egg. "For this reason, we have the extraordinary opportunity to reproduce complex in vivo processes in an in vitro experiment," Baumann said.
Due to its enormous potential, the ZLM helps to reduce the number of animal experiments required, e.g. on rodents and mammals, and thus fulfills a criterion in terms of Reduce, Replace and Refine, also known as the 3R principle. The 3R principle is an important basis for the design and implementation of animal experiments. In this context, the ZLM not only contributes to the reduction of animal experiments as a replacement method, but can also be regarded as a refinement method in particular. "By fertilizing the eggs externally, i.e. outside the mother's body, the embryos and larvae used for the experiments are obtained completely non-invasively without any suffering for the parent animals," explains Jonas Baumann. The model is already used in various research areas at HIPS: in particular in the investigation of the efficacy of drug candidates against human pathogenic bacteria, the investigation of the effects of drugs in the organism (e.g. their metabolism or excretion), or the evaluation of potential toxic side effects. The latter is now also being pursued with Helmholtz colleagues at the CISPA Helmholtz Center for Information Security in a joint collaborative project. Larval development is analyzed with machine learning methods using an automated image-based approach. In this way, new active substances can be tested for their safety at a very early stage, which will accelerate the entire process of drug research.
Last summer, the zebrafish team at HIPS was awarded the research prize "Alternatives to Animal Testing" by the Saarland Ministry of the Environment. Model systems such as the zebrafish larvae model will not be able to completely replace the need for animal testing, but they have great potential to reduce the need for laboratory animals and already play an important role in preclinical drug development.