ADMET
Absorption, Distribution, Metabolism, Excretion and Toxicity
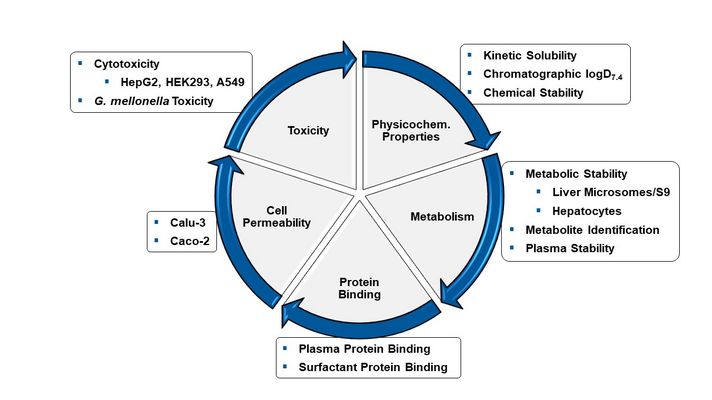
In the translational phase of transferring academic drug-discovery projects into treatment options (from bench-to-bedside), we put a strong focus on the determination of in vitro ADMET properties (absorption, distribution, metabolism, excretion and toxicity). For successful drug-discovery, it is of vital importance to consider these pharmacokinetic properties of a potential new drug candidate early on in a discovery campaign. Towards this end, HIPS has established a rational in vitro screening cascade to analyse ADMET properties such as kinetic solubility/ logD7.4, metabolic stability/plasma stability in diverse species, permeability across cell layers, plasma protein binding and cytotoxicity. The applied technology includes state-of-the-art UHPLC-MS/MS (Triple Quad/Orbitrap), which further allows identification of metabolites.
Continuous screening of ADMET properties is guiding optimisation of compounds in-house as well as in collaboration with external groups. The results generated herein serve as a basis for the selection of compounds for in vivo studies, such as in our in-house zebrafish facility.